Popular Exactech® Knee and Ankle Implants Recalled Due To Alarmingly High-Rates of Failure
What To Do If You Received This Recalled Knee (or Ankle) Implant Between 2004 to 2022
From the desk of:
Andy Bederman, Managing Attorney
To Knee or Ankle Implant Patients:
If you (or a loved one) received a knee or ankle implant between the years of 2004 to 2022, this may be the most important message you will ever read...
Here is why...
In February 2022, knee and ankle device manufacturer, Exactech®, issued a recall for its widely-used knee and ankle replacement implants after investigations revealed alarmingly high-rates of premature wear and complications due to improper packaging of the device prior to surgical use.
The manufacturer, Exactech®, has stated that the recalled knee and ankle implants were packaged in "out-of-specification" vacuum bags that did not meet the strict requirements to prevent excessive oxidation of the polyethylene (plastic) inserts that are critical components of these devices.
Continue reading...

Figure 01 - Diagram of an Exactech® total knee arthroplasty device with the recalled polyethylene insert.
Does This Recall Affect Me?
If you received a total knee or ankle implant between the years 2004 to 2022, the recalled implant may have been used.
The device manufacturer, Exactech®, has reported that 147,732 of the recalled devices have ALREADY been surgically implanted in U.S. patients.

Figure 02 - Data suggests approximately 147,732 recalled devices were implanted in patients in the U.S.
Many total knee arthroplasty recipients are unaware what brand knee replacement they received -- or it's been so long since the surgery that it has been forgotten.
If you don't know what brand knee or ankle implant you received, that's okay. It can easily be determined through either A) medical record retrieval, or B) a simple call to your surgeon's office.
The manufacturer of the recalled device, Exactech®, has recalled all three generations of their knee and ankle systems that have been in use since 2004.
They include, the Exactech®...
- OPTETRAK™
- OPTETRAK™ Logic
- TRULIANT™
- VANTAGE™
Within each generation system, there are multiple polyethylene (plastic) components that were improperly packaged that could lead to significant complications or corrective revision surgery.
The recalled devices are listed below:

Figure 03 - Exactech® Ultra-High Molecular Weight Polyethylene (UHMWPE) Knee and Ankle Polyethylene Inserts packaged in out-of-specification vacuum bags. (from Exactech® website)
Has Exactech® (the device manufacturer) Acknowledged This Problem Exists?
Yes. In fact, Exactech® was the entity that initiated the recall which was classified by the FDA as a Class II Recall.
Exactech® has fully acknowledged that both knee and ankle polyethylene inserts were shipped in improper packaging since 2004.
Further, Exactech® has admitted that numerous studies have shown statistically significant higher-rates of complications with their knee and ankle implants that used these recalled inserts.
Continue reading...

Are My Doctors Aware of This Recall?
If your surgeon ever used the recalled devices, they are likely aware of this recall.
Exactech® has sent letters to all Exactech® knee and ankle surgeons, hospitals and health care professionals explaining the reasons for the recall. Exactech® also provided those surgeons with a draft letter to send to their patients explaining the recall.
You may have even received this letter from your surgeon stating that you, unfortunately, were a recipient of one of the recalled Exactech® devices.
If you haven't received any communications from your surgeon, that doesn't necessarily mean you did not receive a recalled device. So it is always best to confirm by contacting your surgeon's office.
It is also worth noting that your primary care physicians are likely not aware of this recall -- nor aware that you may have been the recipient of a recalled device.
How Do I Determine If I Received The Recalled Implant?
To find out if you were a recipient of a recalled Exactech® polyethylene insert, we suggest taking the following steps as needed...
Call your surgeon's office and ask the following:
"My name is ___________ (YOUR NAME) and I was a patient that received a total ___________ (KNEE or ANKLE) arthroplasty. I would like to know if I received a recalled Exactech® device. If you could kindly let me know the brand, model and serial number of the ___________ (KNEE or ANKLE) system I received and if any of the components have been recalled."
While many doctor offices are quick to get you this information, some can be a bit difficult to get answers right away. So if you have any trouble getting an answer within 1-2 business days, our suggestion is to call every day until you get your question answered.
Continue reading...

The Recall Is Due to "Improper Packaging" -- But What Does That Mean Exactly?
Exactech® total knee and ankle systems utilize a component called a "polyethylene insert" -- which separates the two metal components that are attached to the femur and tibia, respectively.
It is dangerous to have metal rubbing against metal -- as this can cause metal debris to form causing serious infections and complications.
So Exactech® implants avoid this by using a polyethylene (plastic) insert to separate the moving metal components attached to the femur and tibia. The plastic component sits in-between the metal components and allows for smooth movement with low-friction and less wear.
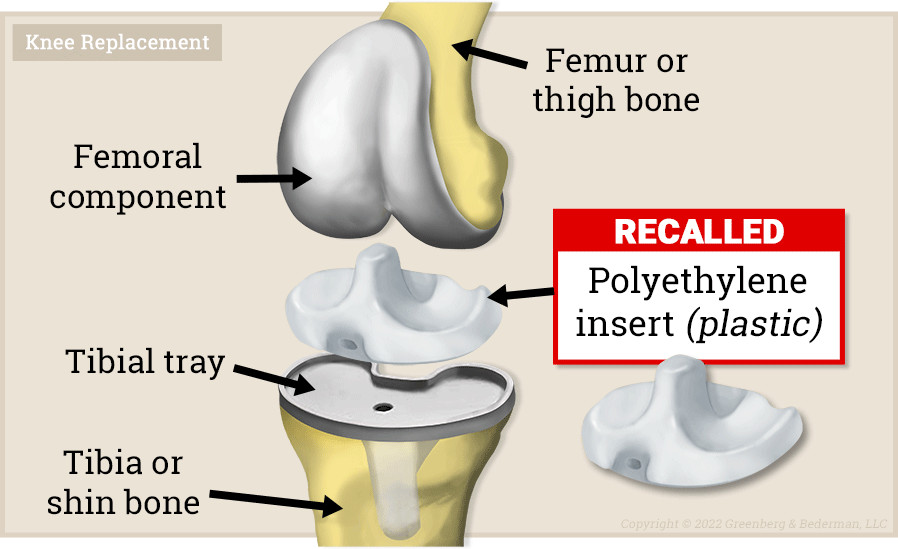
Figure 04 - Diagram of Exactech® knee arthroplasty device with recalled polyethylene insert.
However, polyethylene (a type of plastic) is highly susceptible to oxidation -- which means it degrades over time simply being exposed to air.
As such, it's important for polyethylene components to be packaged in a highly "oxygen resistant" bag. In fact, there are strict requirements set forth by the FDA as to how device manufacturers should package polyethylene devices.
Polyethylene devices that are packaged in vacuum bags (as is the case with Exactech® knee and ankle implants) need to use a secondary barrier layer containing ethylene vinyl alcohol (EVOH). This added barrier layer is what gives the packaging the required oxygen resistance which prevents premature damage to the polyethylene device.
Exactech® admits that MOST of the polyethylene inserts they've manufactured since 2004 were NOT packaged with this required oxygen resistant barrier layer.
In a letter from Exactech® to surgeons, Exactech® stated the consequences of their failure to include this critical and necessary layer of packaging:
"Over time, oxygen can severely degrade the mechanical properties of conventional polyethylene inserts, which, in conjunction with other surgical factors, can lead to both accelerated wear debris production and bone loss, and/or component fatigue cracking/fracture, all leading to corrective revision surgery." -- Exactech® Letter to Surgeons
How Do I Know If My Implant Prematurely Oxidized and Is Damaged?
If you know you received an Exactech® implant, the following complications with your knee or ankle are clues that premature oxidation of the polyethylene insert may have occurred:
- Loosening/instability of the joint
- New or worsening pain
- Inability to bear weight
- Grinding, clicking or other noises
- Swelling
- Osteolysis (bone loss)
- Need for revision surgery
If you've experienced any of the above complications, you may be a victim of Exactech®'s failure to properly package the polyethylene inserts.
What Does Premature Oxidation Look Like?
Premature oxidation of the polyethylene insert can be devastating.
Take a look at the examples below to see the different cases of oxidation with their associated damage.
Premature oxidation can lead to the device producing wear debris, cracking and/or fracturing. All of which would likely require a revision surgery to correct.

Figure 05 - A comparison of polyethylene inserts with various levels of oxidative damage.
Will I Need Revision Surgery?
Only your surgeon can assess the need for a revision surgery to correct any problems that premature oxidation of your insert may have caused.
There are indeed implant patients that have (and will) require corrective revision surgery, however.
Perhaps you have already underwent corrective surgery -- or are scheduled to do so.
Regardless, Exactech® has put out the following statement regarding corrective actions for those patients who received a recalled device:
"For patients who exhibit premature polyethylene wear, the surgeon should consider revision surgery per their clinical judgement."
So please consult with your surgeon, if you have not already done so.
Exactech® Has Asked Me To Call Their "Claims Helpline" -- Should I Call It?
The recalled device manufacturer, Exactech®, has setup a "Claims Helpline" enticing victims of their recalled implants to recover "recall-related out-of-pocket expenses" they may have incurred.
We strongly advise patients to NOT call this claims helpline -- as it is way for the device manufacturer to get you to sign away your rights to sue them for any potential negligence involved in their manufacturing of a defective product.
Instead, we strongly encourage patients who were recipients of the recalled devices to contact an attorney to learn what your legal rights are as a victim of a recalled medical device.
Should I Speak To An Attorney?
When a medical device that you have inside your body is recalled, it is extremely important to protect your rights, your finances and your health.
First, know that you SHOULD NOT feel obligated to file a "claim" through the Exactech® "Claims Helpline". Exactech® would love nothing more than for you to sign away your rights to participate in a nationwide litigation where you could potentially recover significant financial compensation.
Second, you SHOULD speak to a medical device attorney who can explain what typically happens in these kinds of events where a knee or ankle implant is recalled and corrective revision surgery may be needed.
Defective medical devices typically cost an exorbitant amount of money to fix. The revision surgery is expensive, the physical therapy afterwards is expensive, time off work and lost wages can be significant, and so on.
This is where a medical device attorney can help you seek the most financial compensation possible from the at-fault party (the device manufacturer in this instance).
Why Should You Suffer Financial Loss and Emotional Burden Due to Defective Packaging?
As a result of Exactech® (manufacturer of the recalled devices) failing to adequately package their polyethylene inserts, you or a loved one may have suffered complications, needed revision surgeries, physical therapy, experienced emotional distress, incurred lost wages due to time off work, and other significant related expenses.
Because of this, you may be entitled to what are called "damages", which is just a legal term for money awarded in individual lawsuits, settlements and class action lawsuits.
We advise victims of Exactech® implants to strongly consider individual lawsuits and their advantages, compared to participating in a class action lawsuit where typically the damages recovered for each individual claimant are much less.
What Your Next Step Should Be If You Suffered Complications With A Recalled Exactech® Knee or Ankle Implant...
Our legal team has reviewed the available evidence and research, and our experience in medical device liability cases tell us, that we are in prime position to help victims of the Exactech® knee and ankle implant recall and any subsequent complications suffered.
We are offering a free legal consultation and case evaluation to implant recipients who meet the following criteria:
- Received an Exactech® implant between 2004-2022
- Has received (or will receive) corrective revision surgery to remove the implant
If you'd like to learn more and speak with an Exactech® recall lawyer free of charge, this is your opportunity to discover the help that is available to you.
Act Now Because There Is A Limited Window of Time To Pursue A Legal Case
There is a very small window of time for victims to seek financial compensation after an injury -- it is called the "statute of limitations" and the time window for pursuing a case is different in each state.
So please do not wait to speak to an attorney because you do not want to miss your opportunity to pursue an Exactech® lawsuit or an Exactech® settlement.
Getting started is simple and easy...
Here is how to get started:
Fill out the free consultation form below and one of our attorneys will promptly call you to discuss your case.
This consultation is FREE. There is no-risk and no obligation. This is simply your opportunity to understand if you have a case and discover what your legal options are in terms of pursuing financial compensation.
HIGHEST RATINGS FOR ETHICAL STANDARDS AND LEGAL ABILITY
Greenberg & Bederman has achieved the highest possible rating for ethical standards and legal ability by Martindale-Hubbell® for 2022 (and previous years as well). They describe our perfect 5.0 rating as, "a significant rating accomplishment -- a testament to the fact that a lawyer’s peers rank him or her at the highest level of professional excellence and ethical standards."
Additionally, our attorneys have won numerous 2022 Super Lawyers™ Awards.
And know this...
If at any point you do not believe we are the most qualified to help you get the compensation you rightfully deserve, you are not obligated in any way, shape or form to pay us or work with us. This is a NO-PRESSURE consultation.
We are confident you will see the extraordinary value we can offer you. Our law firm works strictly on a contingency basis -- meaning patients we represent only pay out of their recovery if they win their case.
So act now and fill out the contact form at the bottom of the page. Or simply pick up the phone and call us right now at 866-654-8360.
We stand ready to help you win your case.
Sincerely,
Andy Bederman
Owner & Managing Attorney
Law Offices of Greenberg & Bederman, LLC
Get Your FREE Legal Consultation!
Simply fill out the form below and one of our lawyers will promptly call you for your free legal consultation!
Want to call us instead? Call our attorneys at 866-654-8360 right now!
CONTACT US
Silver Spring, MD 20910
WE ARE A NATIONAL FIRM
Accepting Cases In All 50 States
Local: 866-654-8360 (24/7)
Toll-Free: 866-654-8360 (24/7)
Email:
AWARDED SUPER LAWYER™ STATUS IN 2026
The Law Office of Greenberg & Bederman has numerous attorneys that were awarded the prestigious Super Lawyers™ status. Super Lawyers™ is an independent, attorney ranking organization that awards attorneys each year based on many factors, including affinity with other lawyers.
Copyright © 2026 Greenberg & Bederman, LLC.
All Rights Reserved.
Greenberg & Bederman, LLC is associated with Wright & Schulte, LLC
Click to Scroll to Top of Page


