

NuVasive MAGEC® For Scoliosis Linked to Serious Complications
Mounting Reports of Metallosis & Device Failure With This Early-Onset Scoliosis Implant
Complications linked to MAGEC® include...
- Metallosis (metal debris toxicity)
- Premature device fracture (broken implant)
- Necrosis (tissue death near implant)
- Revision surgery to fix failures
- ...and numerous other complications
ALARMING NEWS: Several studies and investigations have recently uncovered significant complications in children implanted with NuVasive MAGEC®. A team of physician-lead investigators followed a group of 30 children implanted with MAGEC® for 18 months and concluded the device has a “substantial complication rate.”
WAS YOUR CHILD A VICTIM OF COMPLICATIONS?
If you think your child may have been affected by such complications, you may be eligible for financial compensation.
Talk to one of our attorneys today. We are offering free legal consultations for a limited time. Click the button below (or call us) to discover how you may benefit...
It's totally free and without obligation.
You may also contact our attorneys at: 866-306-4860
MAGEC®: A Controversial Device For Children With Early-Onset Scoliosis (EOS)...
While NuVasive’s MAGEC® device promises fewer distraction surgeries and a more non-invasive treatment for early-onset scoliosis (EOS), this new implant is causing many concerns amongst health professionals.
Early-onset scoliosis (EOS) is a condition in children characterized by an incorrect curvature of the spine. This deformity can lead to serious structural complications and even crowding of the chest cavity and lungs (thoracic insufficiency syndrome), if left untreated.
Traditionally, early-onset scoliosis (EOS) is treated by implanting “fixed growing rods” to help straighten the spine. But as a child grows, on-going surgeries are required to continually implant longer growing rods -- sometimes subjecting children with EOS to as many as 10 repetitive surgeries.
The NuVasive MAGEC® was introduced to market with the hopes of eliminating these repetitive surgeries -- requiring the child to only undergo a single surgery to implant a MAGEC® growing rod.
Unlike traditional "fixed growing rods" that need replaced, the MAGEC® is unique in that it can be extended using magnets externally -- without the need for invasive surgery.
But researchers and doctors are finding new evidence that the NuVasive MAGEC® system may be causing more harm than intended.
Did your child receive the NuVasive MAGEC® system?


44-Month Multi-Patient Follow Up Study: 6 of 8 MAGEC® Patients Suffered Serious Complications That Required Revision Surgeries
In one of the longest studies of MAGEC® device use, spinal surgeons published a study that showed an alarmingly high incidence of serious complications in patients -- with 6 out of 8 children who received the MAGEC® device requiring revision surgeries.
Eight children with the MAGEC® implant were evaluated for 44-months in this study. Of the eight children with MAGEC®, two suffered complete device fractures and one other child’s device failed due to a part malfunction.
Another child in the study had a device failure that caused the rod to never lengthen -- defeating the whole purpose of the device in the first place.
Only two of the eight children followed for 44-months were able to avoid significant complications requiring numerous surgeries to fix.
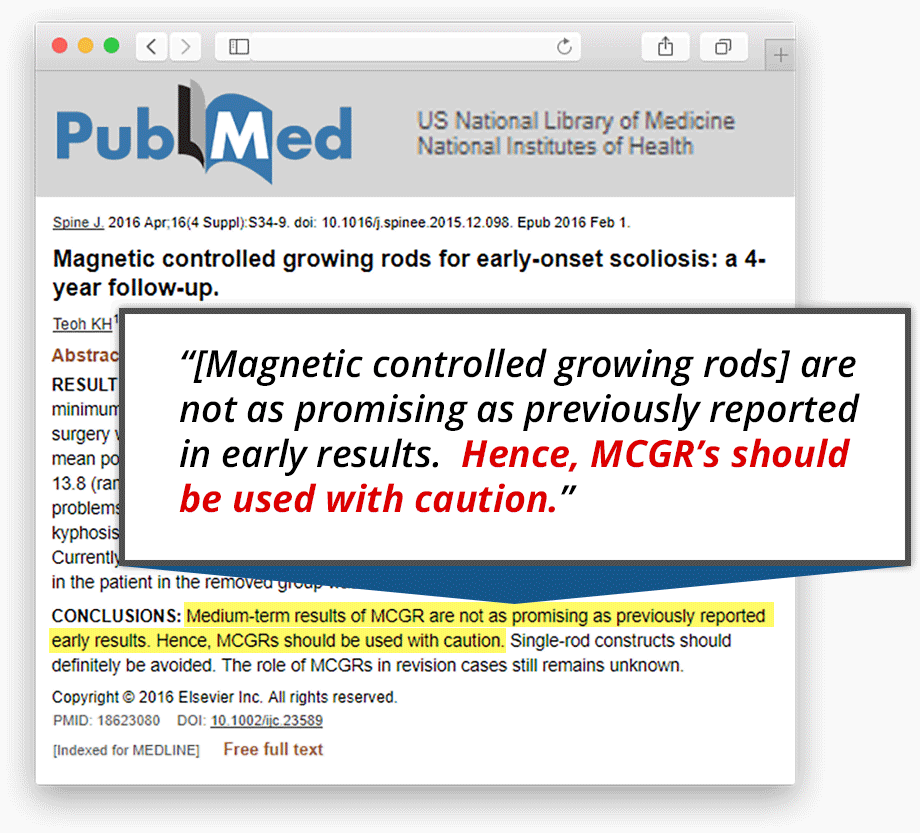
Spinal Surgeons State MAGEC® Implant Isn’t As Promising As Previously Reported...
The spinal surgeons who authored the 44-month study concluded that the medium-term results of magnetic controlled growing rods (ie. MAGEC®) are “not as promising as previously reported in early results.”
And ending in a stern warning, they stated:
“Hence, [magnetic controlled growing rods] should be used with caution. Single-rod constructs should definitely be avoided.”
Was your child a victim of a serious complication with the MAGEC® implant?
FREE LEGAL CONSULTATIONS
AVAILABLE FOR FAMILIES OF MAGEC® PATIENTS
You can talk to our friendly lawyers for free.
You may also contact us anytime at: 866-306-4860
4 out of 5 Patients In Study Suffered From Tissue Metallosis Due to Metal Debris From MAGEC®
Several peer-reviewed studies have emerged demonstrating an overwhelmingly high incidence of metallosis in patients who received the NuVasive MAGEC® implant.
The most recent was a December 2016 study published in The Bone & Joint Journal that evaluated five patients who required revision surgery following the implanting of magnetic controlled growing rods for EOS (ie. MAGEC®).
What they found was astonishing.
Four of the five patients evaluated displayed signs of detrimental metallosis.
The researchers reported that when the implants were opened, “a significant amount of metal debris was found”.
What is Metallosis?
Metallosis is the release of metal debris, and the subsequent build-up of metal debris in the soft tissues of the body.
In the case of MAGEC®, the device is prematurely wearing and causing abrasion -- resulting in the release of titanium alloy debris and particles into the surrounding tissues of the implant, and even into the bloodstream.
This can be an extremely serious and can have permanent, life-altering consequences.
Was your child diagnosed with metallosis or necrosis?


MAGEC® Plagued With High Incidence of Metallosis -- A Highly Troublesome Consequence of Premature Wear & Failure
As explained earlier, metallosis is a serious condition that occurs when a medical implant like the MAGEC® defectively releases metal debris into the surrounding soft tissues, and even into the bloodstream.
The most common symptoms of metallosis are the following:
- Extreme pain around the site of the implant
- Swelling or inflammation
- Loosening or fracture of the implant
- Local necrosis (tissue death)
- Skin pigmentation and discoloration
Necrosis (death of the surrounding tissues due to metallosis), is a particularly dangerous condition that can occur with the MAGEC® implant -- as the loss of tissue (including muscle) can be permanent.
Further, both metallosis and necrosis take a tremendous toll on the child. Usually these conditions require extensive revision surgeries. Not to mention metallosis and necrosis can alter the physical appearance of the child, as skin discoloration and pigmentation is a common side effect of these conditions.
The Law Is Your Protection and Hope:
What To Do If Your Child Has Suffered Complications With The MAGEC® Implant
Medical device manufacturers are bound to strict guidelines and rules set by the FDA.
The FDA puts rules in place that require medical device manufacturers to conduct extensive testing and evaluate continuous research and studies regarding their device's safety so any necessary warnings can be promptly made.
This is done so doctors and the families of children, like your own, can weigh the benefits of using a particular implant against the possible dangers and consequences it may pose.
And it gives doctors the ability to make an informed decision about which type of EOS growing rod to use, based on available safety data gathered in both pre-market and post-market testing and studies.
As a result of NuVasive® failing to properly warn the public of the many known risks associated with the MAGEC® implant, your child and your family has likely suffered significant health complications, revision surgeries, emotional and physical stress, along with unintended financial burdens -- like doctor and hospital visits, time off work, necessary treatments and/or surgeries to correct the failures of the MAGEC® device.
As such, you and your child may be entitled to financial compensation in the form of individual lawsuits, settlements and class action lawsuits.
It’s Worth Speaking to An Attorney:
Get A Free Legal Consultation Today
Our legal team has reviewed the available evidence and our experience tells us that we are in prime position to represent victims who have had serious complications with the NuVasive MAGEC® implant for early-onset scoliosis.
We are currently accepting cases nationally from all 50 states.
For a limited time, our medical device attorneys are offering free legal consultations.
This is your opportunity to have a conversation with an experienced attorney who can answer all your questions and help you determine if you are eligible to pursue financial compensation.
Act Now Because There Is A Limited Window of Time To Pursue A Legal Case
There is a very small window of time for victims to seek financial compensation -- it is called the "statute of limitations" and the time window for pursuing a case is different in each state.
So please do not wait to speak to an attorney because you do not want to miss your opportunity to pursue a NuVasive MAGEC® settlement or lawsuit.
Fill out the form below to begin...
Get Your FREE Legal Consultation!
Simply fill out the form below and one of our lawyers will promptly call you for your free legal consultation!
Want to call us instead? Call our attorneys at 866-306-4860 right now!
CONTACT US
Silver Spring, MD 20910
Local: 866-306-4860 (24/7)
Toll-Free: 866-306-4860 (24/7)
Email:
AWARDED SUPER LAWYER™ STATUS IN 2018
The Law Office of Greenberg & Bederman has numerous attorneys that were awarded the prestigious Super Lawyers™ status. Super Lawyers™ is an independent, attorney ranking organization that awards attorneys each year based on many factors, including affinity with other lawyers.
Copyright © 2026 Greenberg & Bederman, LLC.
All Rights Reserved.
Greenberg & Bederman, LLC is associated with Johnson Becker, PLLC
Click to Scroll to Top of Page


